project cape

hydrogen fluoride
Hydrogen Fluoride (HF), a solution of Anhydrous Hydrogen Fluoride (AHF) in water, is a colorless liquid with a pungent odour.
naturally produced
Fluoride is mostly naturally
occurred in a chemically bonded form, which has a distinctive feature of
releasing Fluoride when Fluorite (CaF2) is grinded. While Fluoride produced
from earth crust accounts for approximately 0.007%, naturally produced Fluoride
compounds mainly exist in the form of natural sediments such as CaF2 and
Cyrolite.
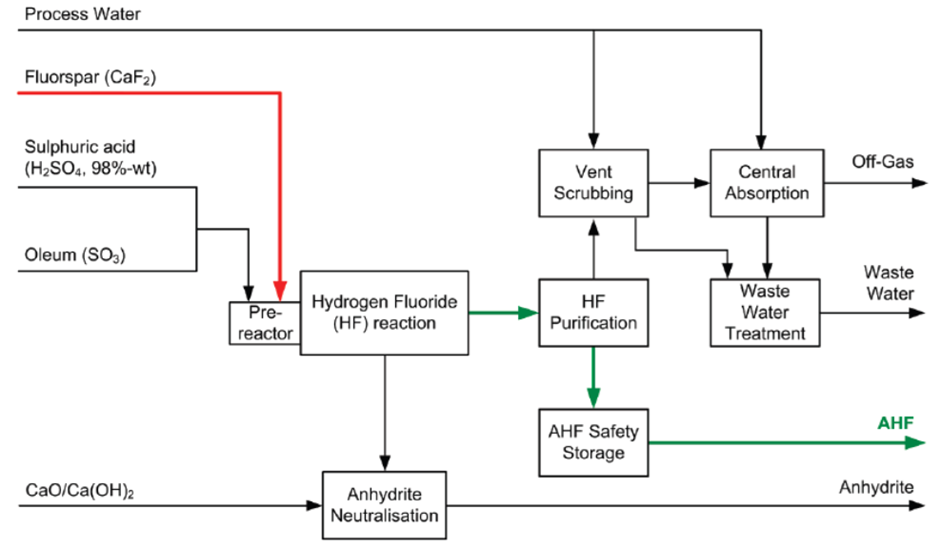
HF Manufacture
HF is first generated in the
form of HF gas by the endothermic reaction of CaF2(grinded/dried) and Sulfuric Acid (H2SO4) within the reactor, which is then purified
and condensed. It is then diluted and absorbed with water to create HF of
desired purity, such as HF 55%, 70%, etc.
chemical reaction
CaF2 + H2SO4 = CaSO4

